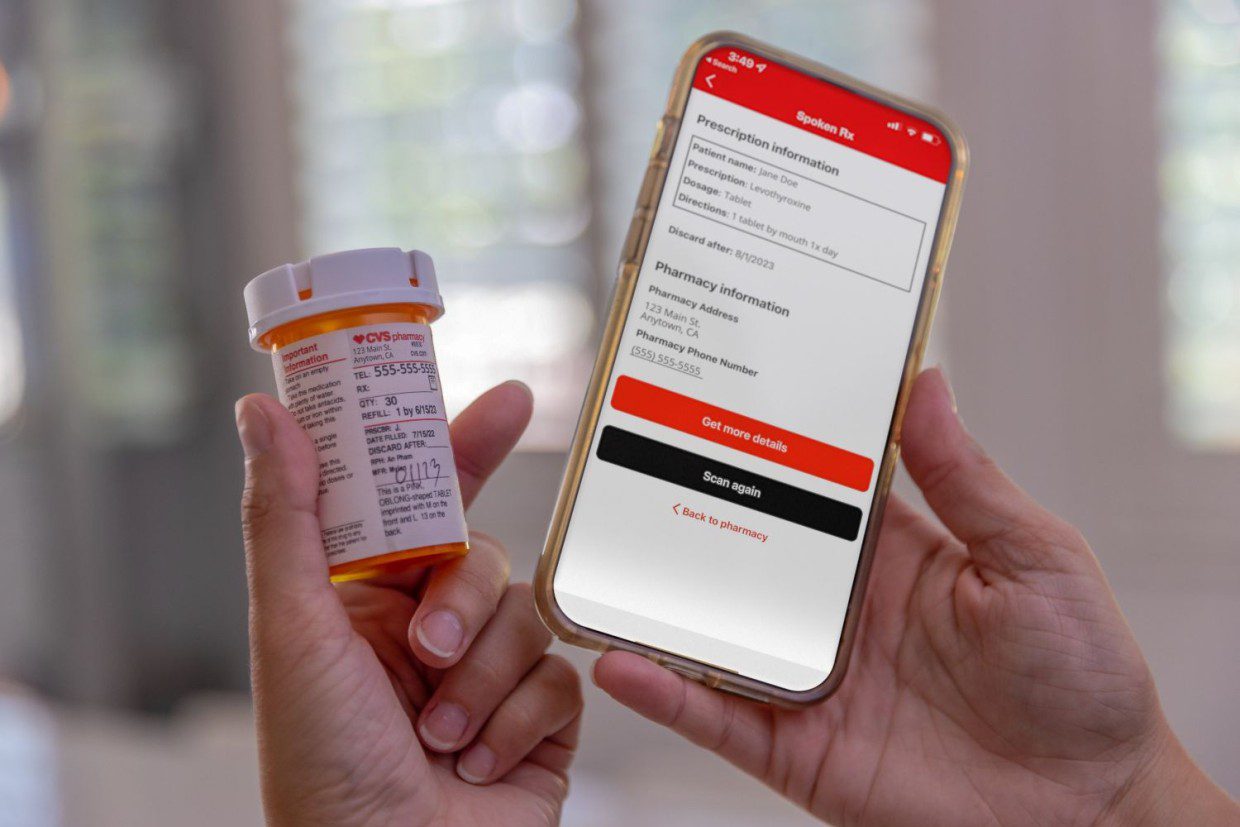
We speak with Deepak Prakash, Vice President, Healthcare at global digital security and identification company, Identiv, about the growing pressure GLP-1 therapies are placing on pharmaceutical cold chains, the risks emerging across last-mile delivery, and why item-level intelligence is becoming essential to maintaining product integrity at scale.
ENABLING PHARMACEUTICAL SUPPLY CHAIN VISIBILITY
As demand for glucagon-like peptide-1 (GLP-1) therapies accelerates and direct-to-patient delivery becomes more common, pharmaceutical cold chains are being pushed into more complex and fragmented distribution models.
What was once a tightly controlled and highly predictable system is now under pressure from increased shipment volumes, more handoffs, and greater exposure to uncontrolled environments, particularly in the final stages of delivery.
For therapies that require strict temperature control and careful handling, this shift is exposing critical gaps in visibility and accountability.
Temperature excursions, handling inconsistencies, and limited oversight in the last mile can all compromise product integrity, while the rise of compounded alternatives is placing even greater emphasis on authentication and traceability at the item level.
Deepak Prakash, Vice President, Healthcare at Identiv, outlines how the company is helping address these challenges through sensor-enabled Bluetooth Low Energy (BLE) smart labels and digital identity solutions that bring continuous, item-level visibility to pharmaceutical logistics.
By enabling products to communicate their condition, movement, and provenance in real time, Identiv is helping manufacturers and logistics providers move beyond retrospective cold chain monitoring toward a more connected, intelligent, and accountable model.

Healthcare Outlook (HO): As the demand for GLP-1 doses continues to surge and the frequency of direct-to-patient delivery increases, what specific vulnerabilities has this shift exposed in the cold chain?
Deepak Prakash, Vice President, Healthcare (DP): The rapid adoption of GLP-1 therapies is reshaping how high-value pharmaceuticals move through the supply chain.
What was once a tightly controlled and predictable system now operates across a broader mix of environments, handoffs, and delivery models, including channels that reach patients directly.
That expansion is being driven by pure scale; JPMorgan projects GLP-1 usage in the US could reach 30 million patients by 2030, with the cold chain packaging market expected to reach $71.6 billion by 2034.
With volume and distribution complexity growing in tandem, the ability to maintain consistent oversight becomes more difficult.
The key vulnerability this exposes is fragmentation – products no longer move through a single, controlled pathway; they pass through a series of independent environments, each with its own workflows.
And as those touchpoints multiply due to GLP-1 demand, so do the chances for inconsistencies in handling and gaps in accountability, making it harder to confidently assess product integrity.
HO: With GLP-1 therapies requiring a strict 2 to 8°C temperature range, how significant is the risk in the final “last mile” environment – in terms of both temperature control and other variables – and what does the industry still underestimate about what happens between pharmacy dispatch and patient receipt?
DP: The last mile introduces more variability and less oversight than earlier stages of the supply chain. Delivery timelines can change, packages may be exposed to fluctuating conditions, and products often sit unattended before receipt.
For therapies with narrow temperature tolerances, even short deviations carry consequences, particularly when they go unnoticed.
The financial scale of cold chain failures reflects how persistent this challenge is. The biopharmaceutical industry loses an estimated $35 billion annually due to breakdowns in temperature-controlled logistics, with many incidents occurring during transit and handling.
What the industry tends to underestimate is how last-mile risk factors converge. Delays, handling variability, and environmental exposure all overlap.
By the time a product reaches the patient, there is often no clear way to verify exactly what it experienced along the way, creating a black box with direct implications for product integrity and patient safety.
HO: As compounded and alternative GLP-1 products enter the market, how is the definition of cold chain accountability evolving beyond temperature control to include item-level authentication and traceability, such as Identiv’s BLE-enabled smart labels?
DP: That black box problem is precisely what is pushing accountability beyond temperature alone.
With more product types entering more distributed channels, knowing that a shipment stayed cold is no longer sufficient; you need to know that the product is genuine, traceable, and intact at the item-level.
Identiv’s BLE-enabled smart labels, with customizable configurations, support this by assigning each unit a unique digital identity to continuously capture and transmit critical data throughout its journey.
This creates a persistent item-level record that travels with the product, enabling verification of condition, provenance, and integrity at critical handoff points, including delivery.
As visibility becomes continuous, accountability evolves from being a compliance function into a dynamic data-driven capability.
The same item-level intelligence that enables traceability also powers predictive, automated, and responsive supply chains, where risks are identified earlier, interventions occur in real time, and the journey of sensitive therapies is continuously optimized from origin to patient.
HO: From Identiv’s perspective, what does true end-to-end visibility actually look like in a modern pharmaceutical supply chain, and how are your technologies helping close the gaps that exist today?
DP: True end-to-end visibility means the ability to follow each unit from origin to patient with a clear and continuous item-level record of its condition, integrity, and chain of custody, with data that can be accessed and acted on in real time without relying on retrospective reconstruction.
One of the primary challenges in achieving this is interoperability. Information is captured at multiple stages, but it often resides in disconnected systems that don’t communicate effectively.
Without consistent data exchange between platforms, it becomes difficult to assemble a complete and reliable view of a product’s movement.
Identiv addresses this by anchoring visibility at the source. Sensor-enabled labels capture temperature, humidity, movement, and handling in real time, and through collaborations with partners like Tag-N-Trac, that information can be integrated into cloud-based platforms designed to convert raw data into value for specific applications, such as cold-chain.
This makes it accessible across both transit and storage environments as events occur.
For GLP-1 therapies specifically, this enables teams to confirm product integrity in real time and respond before issues reach the patient.
HO: Looking ahead – and as continuous condition monitoring becomes increasingly pertinent – where is Identiv seeing the strongest demand for innovation? Temperature monitoring, authentication, or integrated solutions that combine both?
DP: Integrated solutions, without question. The conversations we’re having are no longer about solving for temperature or authentication in isolation; organizations want a complete picture at the item level. Where did this product originate? How did it move? Was it ever compromised?
Delivering that level of visibility becomes more complex as these therapies move through highly distributed supply chains.
With GLP-1 access expanding, products are passing through more partners and environments, increasing the number of handoffs and potential points of risk.
In response, we’re seeing growing interest in layered security models that combine overt and covert protections; overt features enabling fast and intuitive authentication as products change hands, and covert elements providing a deeper layer of security tied to each unit’s digital identity to guard against diversion, tampering, and counterfeiting throughout the journey.
Together, these approaches enable a coordinated end-to-end accountability model that connects first-, middle-, and last-mile distribution, built to support pharmaceutical access at unprecedented scale.
Sustainability is also emerging as an important driver of innovation. When temperature-sensitive products fall outside required conditions, they are discarded, and that waste accumulates quickly.
It’s been estimated that up to 50 percent of vaccines are wasted globally, with cold chain failures being a major contributing factor.
Real-time insight into product movement and handling enables earlier intervention, helping curb avoidable loss and supporting the move toward more efficient, circular supply chains.
This article was contributed by a guest author and published by the editorial team at Healthcare Outlook, part of the Outlook Publishing global network of B2B industry magazines.
Outlook Publishing features leadership insights, industry perspectives, and company stories from organisations shaping sectors including healthcare, manufacturing, supply chains, construction, mining, food production, and sustainability.
Healthcare Outlook explores the organisations, innovations, and leaders shaping the future of healthcare systems worldwide.